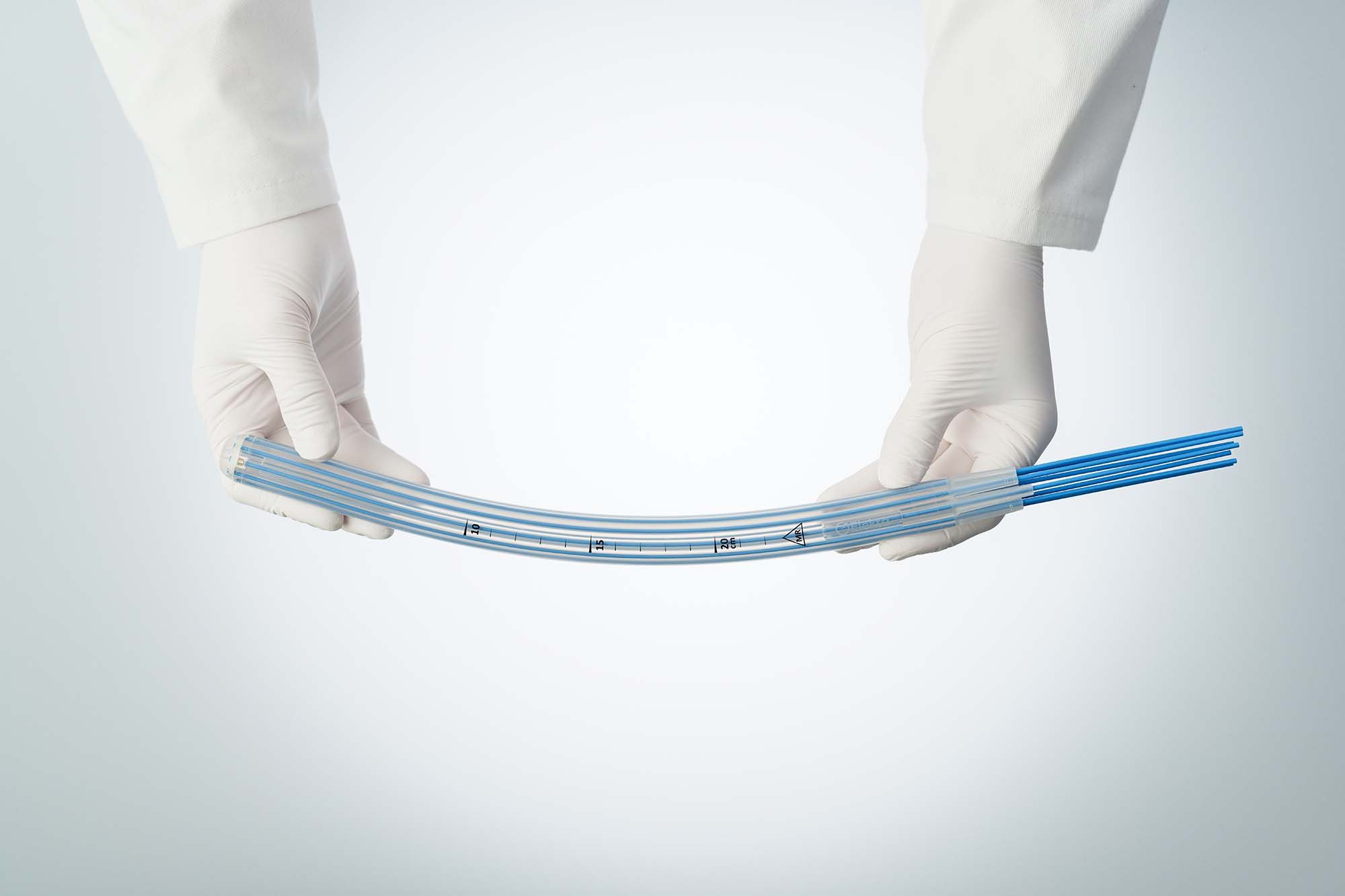
Designed to simplify endorectal brachytherapy treatment
Evidence–backed rectal HDR supporting organ preservation
Seamless integration into existing HDR workflows
Single-use design for enhanced patient safety
We want to ensure optimum use of our website for you, and to continually improve our website. Therefore, we work with selected partners (e.g. Pardot, Google Analytics, Matomo). You can revoke your voluntary consent at any time. You can find further information and setting options under "Configure" and in our data protection information.

Enabling image-guided HDR brachytherapy in rectal cancer
Evidence–backed rectal HDR supporting organ preservation
Seamless integration into existing HDR workflows
Single-use design for enhanced patient safety

Offers maximum flexibility to minimize discomfort for patients during the procedure versus a rigid applicator, especially for elderly and frail patients.


Designed to make assembly of the Fixation Element simple, no screws or screwdriver is needed. The Fixation Element and Fixation Clip, as well as the Insertion Tool, are reusable and can be sterilized by steam sterilization.
The applicator is suitable for use in an MR environment, and provides good imaging results without artifacts or distortion when used with x-ray and CT. In addition, the radiopaque catheters help users to locate twists during reconstruction.

HDR endorectal brachytherapy is an important component of modern rectal cancer treatment, especially in organ preservation strategies.
HDR brachytherapy is used as a dose-escalation boost in combination with chemoradiotherapy (CRT), supporting treatment strategies aimed at improving tumor response and enabling organ preservation in selected patients.
Organ preservation approaches seek to avoid radical surgery while maintaining oncological outcomes and quality of life. HDR brachytherapy contributes to this goal by delivering a highly conformal dose directly to the tumor while sparing surrounding healthy tissue.
Clinical evidence supports the role of HDR brachytherapy in this setting. The MORPHEUS Phase II–III randomized trial demonstrated improved total mesorectal excision (TME)-free survival with an HDR brachytherapy boost compared with an external beam radiotherapy (EBRT) boost (76.6% vs 38.6% at 2 years, interim analysis), with acceptable toxicity1. These findings support its use within organ preservation strategies.
Organ preservation strategies incorporating endorectal HDR brachytherapy are now reflected in clinical guidelines. The European Society for Medical Oncology (ESMO) 2025 guidelines for localized rectal cancer recommend that, for patients with T1–T2 N+ or T3 N0–1 tumors <5 cm in the middle or lower rectum, CRT combined with dose escalation using endorectal brachytherapy may be considered as a treatment option (Level I evidence)2.
Ongoing studies, including MORPHEUS and ACO/ARO/AIO-223, continue to strengthen the clinical evidence base, particularly in populations such as elderly and frail patients, for whom organ-preserving approaches are especially relevant.
Not only has HDR brachytherapy proven to be a safe, effective treatment, it can also be – with the right tool – an exceptionally simple, straightforward therapy. Elekta’s new Rectal Applicator offers you a way to deliver endorectal HDR brachytherapy, facilitating a streamlined workflow. This applies not only if your center has never done brachytherapy for any indication before, but also if you have a well-established GYN and/or prostate HDR brachytherapy service. This single-use applicator is compatible with Elekta’s Flexitron and microSelectron afterloader systems.
References
Applicator insertion
Imaging
Treatment Planning
Treatment Delivery
Applicator Disposal

Products may not be available in all markets
Do you connect from ___?
Due to regulatory restrictions, this content is restricted to some locations. Get in touch with us to learn more about the device availability.
Rectal Applicator is processing CE mark and not available commercially.