Standardizing cervical cancer brachytherapy in India

Multi-institutional study establishes comprehensive guidelines for HDR brachytherapy practice and techniques

Cases of cervical cancer can be found in all parts of the world, but the incidence of the disease is much greater in LMIC’s (low and middle-income countries, e.g., India, Nepal, African nations) than in Western countries. Of GLOBOCAN’s 2018 estimate of 570,000 new cervical cancer cases, 85 percent were diagnosed in LMICs, with half of these individuals dying from their disease. India, in particular, recorded approximately 100,000 new cervical cancer patients, with nearly two-thirds (65,000) dying. As a critical modality in treating cervical cancer, high dose rate (HDR) brachytherapy (BT) has never been more important. The problem in India and other LMIC’s, however, has been wide variability in HDR brachytherapy practice.
“Treatment and in particular brachytherapy techniques are affected by socioeconomic factors, in addition to the availability of human and technological resources, all of which result in very heterogeneous patterns of care,” says Umesh Mahantshetty, MD, Radiation Oncologist at Mumbai, India’s Tata Memorial Centre.
For example, Dr. Mahantshetty says, LMIC’s east of India tend to use HDR BT with very low doses per fraction, delivering multiple treatments over just a couple of weeks. In more western LMIC’s – India and west to the Middle East – the challenge is patient compliance. Getting patients to travel to and stay at a distant center for eight weeks of treatment is difficult. In addition, depending on available resources, there is wide variability in the type of imaging modality that clinics in LMIC’s use for cervical cancer staging, with most centers lacking gold standard MRI or PET/CT. Major inconsistencies also exist in reporting practices and in the use of BT applicators.

“If imaging, reporting and applicator use were more homogenous among LMIC’s – and if centers could practice cervical cancer HDR brachy using standardized guidelines – then after two to five years, we could combine the data among many centers and acquire more reliable information on patient outcomes. This would then help in the development of protocols to improve outcomes,” he says.
Recognizing the opportunity, Dr. Mahantshetty and clinicians at nine other Indian centers that use HDR brachytherapy to treat large numbers of cervical cancer patients, developed a report that reviews, discusses and proposes practical recommendations to ensure minimum standards of radiotherapy with a particular focus on BT in India.1
To create the report, the panel of researchers – all members of the Indian Brachytherapy Society – conducted a literature review and defined guidelines based on available evidence and its relevance to Indian settings. The guidelines and recommendations are exhaustive, covering:
- Pre-treatment and staging workup
- External beam radiotherapy
- Chemotherapy
- Patient preparation and counseling
- Brachytherapy
- Principles and technique of brachytherapy application (intracavitary; intracavitary + interstitial; interstitial)
- Patient eligibility
- Target and organs-at-risk definitions
- Anesthesia
- Applicator reconstruction
- Imaging for treatment planning
- Reporting
- Applicator selection
- Follow-up
Among these components of the HDR brachytherapy protocol, Dr. Mahantshetty identified three presented in the article as particularly noteworthy: pre-treatment and staging workup, principles and technique of brachytherapy application and applicator selection, and brachytherapy complications and how to address them.
Pre-treatment and staging workup
In addition to the standard post-diagnosis tests, including blood tests, renal and liver function assessments and coagulation parameters, Dr. Mahantshetty stresses the importance of clinical drawings for properly documenting local disease topography. Figures 1 is a modification of the EMBRACE group’s clinical drawings, showing hypothetical disease documentation.
“Clinical drawings really make the pre-treatment examination objective and help to show what is happening in the disease process.”
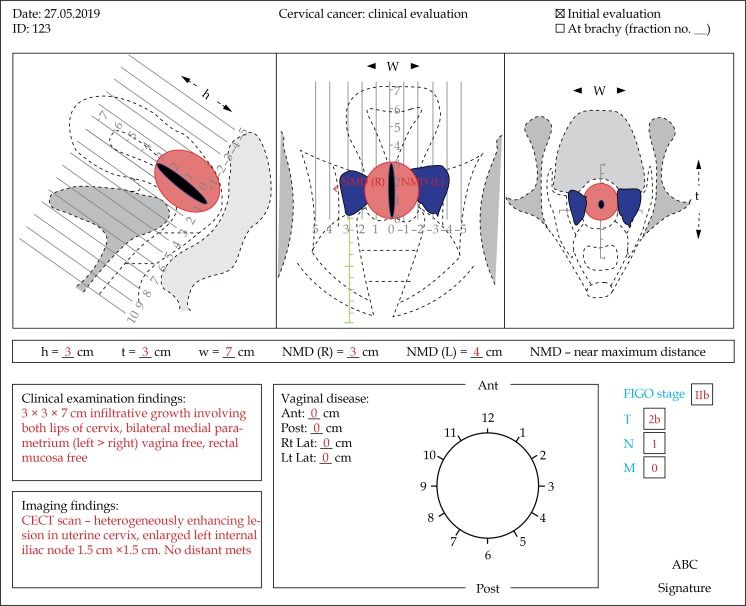
“Clinical drawings really make the pre-treatment examination objective and help to show what is happening in the disease process,” Dr. Mahantshetty says.
Principles and technique of brachytherapy application and applicator selection
Selecting the appropriate BT application technique depends on the topography of the residual disease at the time of BT. Patients are categorized as those with an intact uterus and post-hysterectomy patients, and for both groups the disease is classified according to its extent, i.e., ranging from no residual disease to advanced spread to the cervix, parametrium and vagina. BT techniques are categorized into intracavitary BT (ICBT), combined ICBT and interstitial BT (ICIS) and interstitial BT (ISBT). The Mahantshetty, et al report explains the circumstances under which each technique should be used and describes the various applicators that clinicians can employ.
“Thanks to BT’s development over the years, today we have a large variety of applicators for a range of disease processes.”
“When I was a medical resident 20 years ago, the variety of applicators for treating cervical cancer was very limited, due mainly to the lack of imaging technology to clearly visualize the disease,” he says. “Thanks to BT’s development over the years, today we have a large variety of applicators for a range of disease. The use of vaginal applicators depends on the residual disease in the vagina; the upper vagina is typically well covered with ring/ovoid systems and for extensive vaginal residual disease beyond the upper vagina, vaginal cylinders are preferred.”
An applicator that is especially notable according to Dr. Mahantshetty is Elekta’s Venezia™ advanced gynecological applicator, which he uses at Tata Memorial Centre. Venezia (Figure 2).
Venezia, which belongs to a new class of hybrid (i.e., intracavitary/interstitial) GYN applicators, simplifies the treatment of locally advanced cervical cancer by reducing the need for either perineal template or free-hand placement of interstitial needles.
“I think it’s a good idea that Elekta is promoting Venezia as an advanced gynecological applicator, one that can be used across all the stages of cervical cancer,” he says. “It is designed not only to treat bulky IB and II tumors, but also IIIA and IIIB stage disease. We can even treat patients with IVA disease in which the bladder and/or rectum is involved.”

Brachytherapy complications and how to address them
Because complications are exceedingly rare in HDR BT for cervical cancer, information regarding how to deal with them is sparse, according to Dr. Mahantshetty.
“There have been no articles I’m aware of that address how to tackle complications,” he adds. “More research needs to be done on this subject.”
- Perforation: typically seen as a false passage through the fornices, missing the uterine canal or perforation through the wall of the uterus or cavity. This complication can be prevented by the use of real-time, transabdominal ultrasound intraoperatively during BT. In the event of perforations through the fornices or anterior/posterior walls of the uterus, BT should be abandoned and a procedure under ultrasound guidance is recommended.
- Bleeding: Clinicians may encounter bleeding during BT either from the tumor or as a consequence of applicator removal. In most cases, bleeding can be stopped with vaginal gauze packing and antifibrinolytics. Significant blood loss can be addressed by blood transfusions. If the bleeding was caused by a vaginal tear, repair by suturing should be done under anesthesia.
- Vaginal injuries: BT-related mucosal abrasions and tears are addressed by suturing of the tear and delaying BT for a week to 10 days and antibiotic therapy.
- Bladder/rectal/bowel injuries: A very rare occurrence that may require the care of a surgeon.
An enduring need for HDR brachytherapy
Despite the widespread use of HPV vaccines in most regions of the world, the incidence of cervical cancer, especially in LMIC’s such as India, is steadily decreasing, therefore patients needing treatment for cervical cancer is still going to be a major concern, according to Dr. Mahantshetty.
“The projections from WHO and the International Atomic Energy Agency indicate that cervical cancer incidence will not substantially reduce in next 50 years at least.”
“The projections from WHO and the International Atomic Energy Agency indicate that cervical cancer incidence will not substantially reduce in next 50 years at least,” he says. “Given that, it’s unlikely that the need for HDR brachytherapy will abate.”
With appropriate resources and expertise, more centers in India should consider implementing an HDR brachytherapy program, Dr. Mahantshetty says.
“They should not be intimidated about starting such a program. Clinicians just need to be careful and cautious in implementing all the processes outlined in our paper through diligent quality assurance and quality control,” he says. “Apart from using the article as a blueprint for processes, radiotherapy staff should attend workshops that include hands-on demonstrations and consider taking short courses, such as Elekta’s BrachyAcademy workshops for more insight. These are quite useful in terms of practical tips and tricks in launching a BT program.
“Inviting an expert on site to help guide them for the first few applications also is a great idea,” he adds. “This can increase the confidence level of the whole brachytherapy team very rapidly.
For centers that already practice BT for other indications, implementing an HDR BT program for cervical cancer can be a fast transition. They just need to tweak the processes, but, again, attending workshops using phantoms and cadavers to get the hands-on experience necessary, will help them efficiently implement the program.”
HDR Brachytherapy for cervical cancer at Tata Memorial Centre
- 2 centers: Tata Memorial Centre (main campus) and Advanced Centre for Treatment, Research and Education in Cancer (ACTREC)
- 3 HDR brachytherapy afterloaders (microSelectron® [Elekta])
- 7 applications per day on average (range: 5-12)
- Clinic hours: 8:00 am to 7:00 pm
- Length of time for each patient (all procedures): 90 minutes to 2 hours
1. Mahantshetty U, Gudi S, Singh R, et al. Indian Brachytherapy Society Guidelines for radiotherapeutic management of cervical cancer with special emphasis on high-dose-rate brachytherapy. J Contemp Brachytherapy 2019; 11, 4: 293-306. DOI: https://doi.org/10.5114/jcb.2019.87406