MR guidance and hypofractionated RT for prostate cancer a good match
Wisconsin and Iowa medical centers add to wealth of clinical evidence on safety and value of moderately- and ultra-hypofractionated radiotherapy
Clinicians at the Froedtert & the Medical College of Wisconsin Clinical Cancer Center at Froedtert Hospital and the University of Iowa (UI) are using their respective center’s Elekta Unity MR-Linac system in ongoing efforts to validate the equivalence of hypofractionated prostate cancer regimens to standard fractionation schedules. Although data on the late toxicity of hypofractioned radiotherapy will take time to assess, acute toxicity has been low in the Froedtert & MCW Clinical Cancer Center’s and UI’s experience, while patients benefit from a treatment course shortened by several weeks.
Long before the Froedtert & MCW Clinical Cancer Center acquired its Elekta Unity system, it was involved in National Cancer Institute (NCI) clinical trials evaluating moderate hypofractionation using standard treatment systems.
“Virtually all of the trials that we participated in have shown that you can easily go to moderate hypofractionation and do it safely.”

“It has been an evolution and a process over the last eight to 10 years in NCI trials,” says Colleen Lawton MD, Vice Chair, Department of Radiation Oncology at the Medical College of Wisconsin (Milwaukee, Wisconsin). “Virtually all of the trials that we participated in have shown that you can easily go to moderate hypofractionation and do it safely. And a shorter period of time is good not only for the patients – who don’t have to come to the hospital as often – it’s also good for the health care system, because it’s less costly.”
The results from clinical trials the Froedtert & MCW Clinical Cancer Center has been involved in – in addition to those of several other multi-center studies – have enabled centers to reduce the standard fractionation of 75.6 Gy delivered in 42 fractions over 8.5 weeks to moderate hypofractionation of 70 Gy delivered in 28 fractions over 5.5 weeks or 60 Gy in 20 fractions over four weeks on conventional linear accelerators using cone-beam CT imaging.1-5
The Froedtert & MCW Clinical Cancer Center’s acquisition of Elekta Unity in 2019 changed the imaging equation of prostate radiotherapy dramatically. It provided pristine, MR-quality visualization of anatomy compared to CT, thereby improving the delineation of the prostate. In addition, it gave clinicians the ability to create treatment plans on the MR-Linac, enabling the treatment to be shaped to the patient’s anatomy in the treatment position just before therapy delivery.
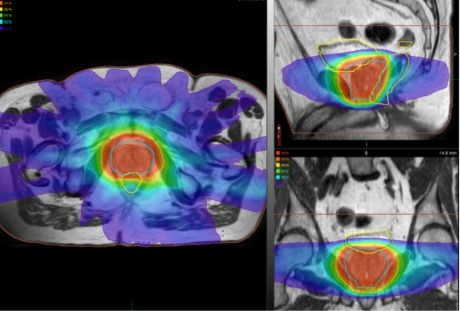
“Being able to modify the treatment just before beam delivery is a big advantage over the standard radiotherapy workflow, which just takes an anatomical snapshot during simulation,” she notes. “That could be two weeks before treatment, and a lot can change anatomically in that time. With Elekta Unity, we’re also able to watch the image during beam delivery, which is important in prostate radiotherapy because the gland can be displaced by bladder and bowel contents during the fraction.”
With Elekta Unity, the quality of the planning images and real-time imaging during beam delivery enable Froedtert & MCW Clinical Cancer Center clinicians – for select prostate patients – to use a moderately hypofractionated schedule of 60 Gy delivered in 20 fractions over four weeks [i.e., PRISM protocol6].
Ultra-hypofractionated SBRT for prostate cancer
The Froedtert & MCW Clinical Cancer Center is participating in an NCI randomized phase III study (NRG-GU005) exploring how well SBRT works versus moderately hypofractionated IMRT in treating patients with stage IIA-B (intermediate risk) prostate cancer7.
The study is designed to determine whether SBRT (five fractions of 7.25 Gy) is superior to moderately hypofractionated IMRT (28 fractions of 2.5 Gy) in terms of patient-reported side effects.
“In general, we think the outcomes of SBRT will be equivalent to moderate hypofractionation, but we don’t have the randomized data yet to prove this,” Dr. Lawton says. “We need these prospective, randomized trials to verify what we think – that a high-dose per fraction, ultra-hypofractionated treatment is equally safe and equally effective.”
The Froedtert & MCW Clinical Cancer Center will be using the Elekta Unity for the patients in this study who randomize to the SBRT arm to take advantage of the system’s superior soft-tissue visualization, particularly its real-time imaging capability.

“Especially with SBRT that uses these very high-dose fractions, Elekta Unity enables us to actually watch bowel gas come through and stop beam delivery if it looks like things are really moving,” she says. “Even in a situation in which you’re using implanted fiducial markers with a conventional system, deformation can occur in the prostate where the fiducials may not change a lot, but the actual outside of the gland has moved. You won’t be able to see that except on a system like Elekta Unity.”
Moderate hypofractionation from the start
For prostate cancer patients at the University of Iowa (UI, Iowa City, Iowa), clinicians have been using their Elekta Unity system since May 2019 to deliver a 60 Gy treatment over 20 fractions, replacing the standard fractionation schedule of 78 Gy delivered in 39 fractions.
“Starting with the very first patient, we adopted the PRISM protocol,” says Mark Smith, MD, UI Clinical Professor and Radiation Oncologist. “We will use the MR-Linac for any patients with low or intermediate-risk disease – those with organ-confined prostate cancer plus the seminal vesicles – but even some patients with high-risk disease for whom we don’t believe it’s necessary to treat the pelvic lymph nodes.”
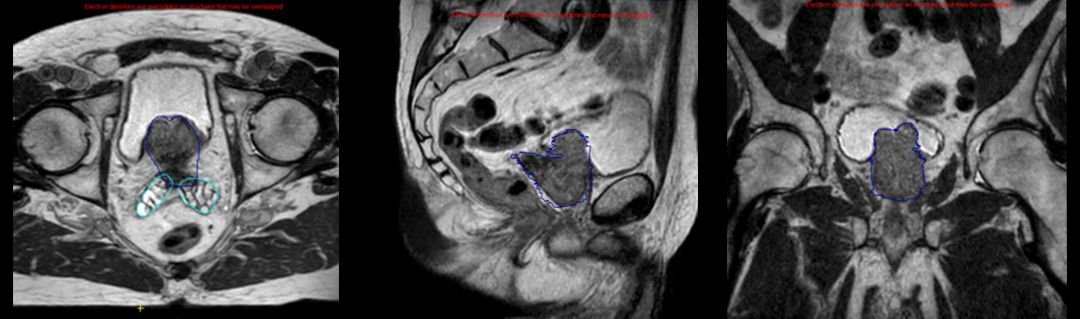
By late September 2020, UI had treated 27 prostate cancer patients with this protocol, representing two-thirds of the center’s volume of prostate patients receiving radiotherapy for their disease. Elekta Unity has also been used for oligometastatic disease and for tumors in other anatomies, such as the liver and brain.
“The trend is definitely in the direction of hypofractionated prostate cancer regimens, because they do appear to be equivalent to traditional long-course radiotherapy.”
“The trend is definitely in the direction of hypofractionated prostate cancer regimens, because they do appear to be equivalent to traditional long-course radiotherapy,” he says. “It’s nice for our patients if you can decrease the number of times they come in and still have comparable results. It’s a win for everyone.”
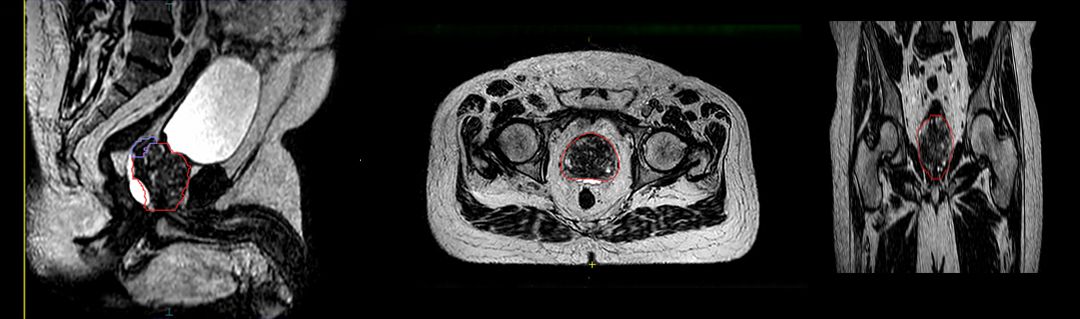
Dr. Smith predicts that Elekta Unity will allow UI clinicians to make the leap from moderately hypofractioned radiotherapy to “true” hypofractionated radiotherapy as soon as next year.
“The key is having the right tools to make sure you can do it safely,” he says.
Learn more about Elekta Unity.
References
- Morgan SC, Hoffman K, Loblaw DA, et al. Hypofractionated radiation therapy for localized prostate cancer: An ASTRO, ASCO, and AUA evidence-based guideline. Journal of Clinical Oncology 2018 36:34, 3411–3430.
- randomised, non-inferiority, phase 3 CHHiP trial. Lancet Oncol. 2016 Aug;17(8):1047-1060. doi: 10.1016/S1470-2045(16)30102–4. Epub 2016 Jun 20. Erratum in: Lancet Oncol. 2016 Aug;17 (8):e321. PMID: 27339115; PMCID: PMC4961874.
- Lee WR, Dignam JJ, Amin M, et al. NRG Oncology RTOG 0415: A randomized phase III non-inferiority study comparing two fractionation schedules in patients with low-risk prostate cancer. Journal of Clinical Oncology 2016 34:2_suppl, 1-1.
- Incrocci L, Wortel RC, Alemayehu WG, et al. Hypofractionated versus conventionally fractionated radiotherapy for patients with localised prostate cancer (HYPRO): final efficacy results from a randomised, multicentre, open-label, phase 3 trial. Lancet Oncol. 2016 Aug;17(8):1061–1069. doi: 10.1016/S1470–2045(16)30070–5. Epub 2016 Jun 20. PMID: 27339116.
- Brand DH, Tree AC, Ostler P, et al. Intensity-modulated fractionated radiotherapy versus stereotactic body radiotherapy for prostate cancer (PACE-B): acute toxicity findings from an international, randomised, open-label, phase 3, non-inferiority trial. Lancet Oncol. 2019 Nov;20(11):1531–1543. doi: 10.1016/S1470–2045(19)30569-8. Epub 2019 Sep 17. PMID: 31540791; PMCID: PMC6838670.
- Dunlop A, Mitchell A, Tree A, et al. Daily adaptive radiotherapy for patients with prostate cancer using a high field MR-linac: Initial clinical experiences and assessment of delivered doses compared to a C-arm linac. Clin Transl Radiat Oncol. 2020;23:35-42. Published 2020 Apr 29. doi:10.1016/j.ctro.2020.04.011
- https://clinicaltrials.gov/ct2/show/NCT03367702