Spinal cord sparing in SABR treatment of vertebral metastasis
Case: Prostate cancer with T5 vertebral metastasis.
Contributor: Vincent Khoo, Arun Jaganathan, Semir Fazlic, Dominika Oborska-Kumaszynska and Henry Weatherburn, Department of Physics, Cancer Centre London, Wimbledon, United Kingdom.
Overview
An 80-year-old male patient with prostate cancer (T4N1M1) presented with PSA 35-48, Gleason 4-5 (6/6 scores positives, 60-90%). MRI showed T4 involvement in the bladder base, N1 bilateral pelvic nodes, possible T5 metastatic lesion and a small right renal mass. Choline PET-CT confirmed a T5 vertebral metastasis in the thorax region with bilateral pelvic lymph node involvement (Figure 1) but no avidity in the renal mass. The renal mass was not related to the prostate cancer and it was an incidental finding that is being monitored.
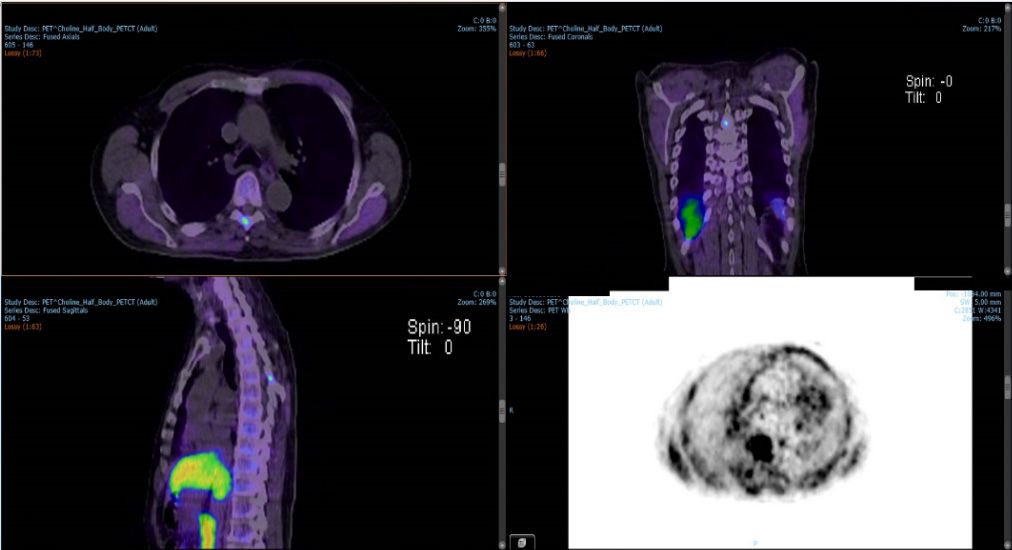
Stereotactic ablative body radiotherapy (SABR) was chosen for treatment of the vertebral metastasis after radical irradiation of the prostate and pelvic nodes. Planning aims were to deliver high-dose spatial resolution (three levels of prescribed dose), incorporating steep gradients between dose levels and at the boundaries between the treated area and OARs/surrounding tissue, while meeting dose constraints for the spinal cord 1-4,8,9. The T5 was not treated in isolation but within the context of the prostate and pelvis nodes also receiving radical RT.
Treatment planning
Treatment planning was performed in Monaco version 5.11.02. The simulation CT and PET-CT scans were fused using ROI registration. The SUV (PET segmentation) function in Monaco was used to enhance visibility and improve the assessment of the “hot” region within the vertebra. OARs and target volumes were delineated according to SABR consortium guidelines (v6.11)1. A multicriterial optimization treatment plan was generated to achieve a suitable three-level dose distribution for SABR treatment of the vertebral metastatic lesion2,7-9.
CTV1 (vertebral body) and CTV2 (posterior vertebra) were expanded to PTV1 and PTV2 respectively, with a uniform margin of 2 mm. A simultaneous integrated boost (SIB) was applied to CTV3 (the volume of high uptake within the vertebra), which formed PTV3 with 0 mm margin (Figure 2). To restrict spinal cord proximity, PTV_Prescribe and PRV_Cord were established, as stipulated by SABR Guidelines1.
A 6 MV high dose rate volumetric modulated arc therapy (VMAT) plan for Versa HD™ was generated to deliver 24 Gy to PTV1, 27 Gy to PTV2 and 30 Gy (boost) to PTV3 in three fractions. For the median prescription doses (30, 27 and 24 Gy), isodose prescription lines were 87%, 78% and 72%, respectively. The plan included four full VMAT arcs (Figure 3). Critical structures to be avoided and dose constraints are shown in Table 1. The maximum doses to the spinal cord and spinal cord PRV (2 mm margin) were 17.81 Gy and 21.9 Gy to 0.1 cc, respectively, which met SABRCG tolerances1,2,4,5.
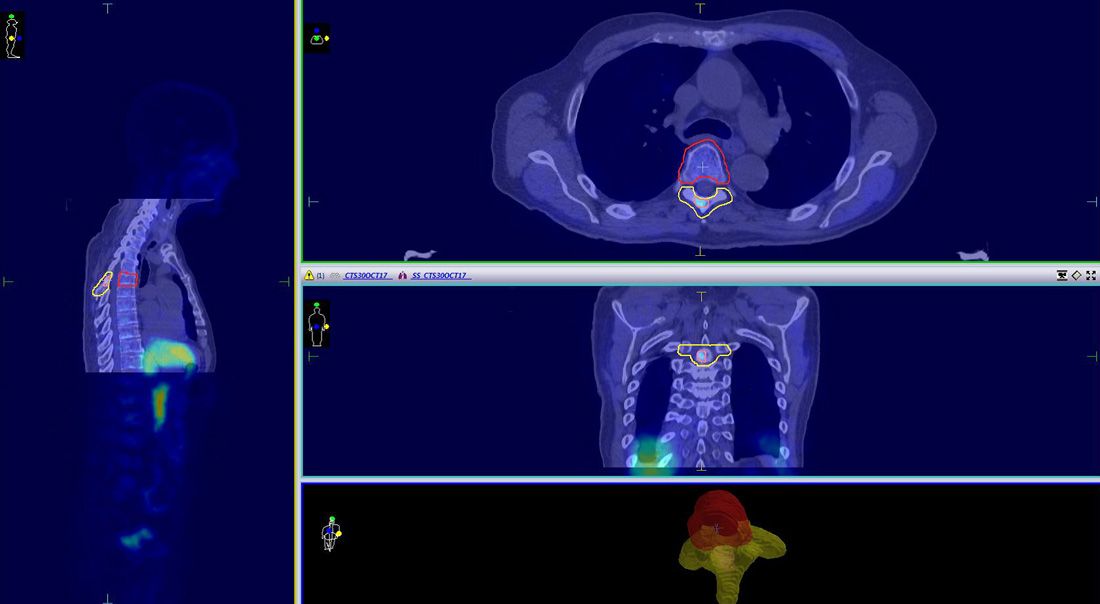
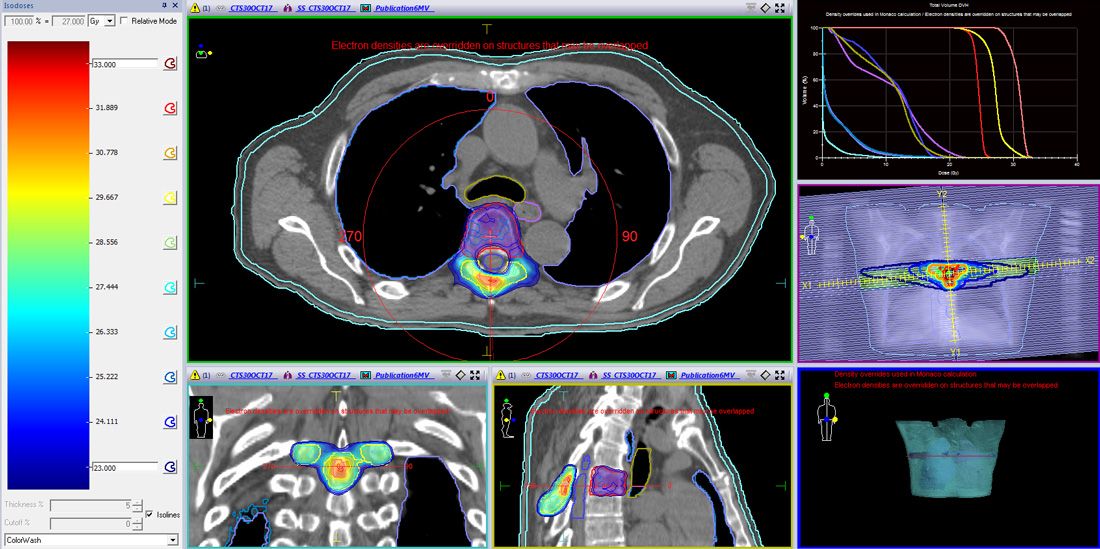
Table 1. Critical structures, dose constraints and mean target doses.
| Critical structure | Criteria | Achieved dose constraint (Gy) |
| Spinal cord | 17.38 (0.1 cc) | |
| Spinal cord PRV | Optimal <18.0 Gy/0.1 cc1 Mandatory <21.9 Gy/0.1 cc1 | 21.91 (0.1 cc) |
| Esophagus | Mandatory <25.2 Gy/0.5 cc1 | 10.68 (Dmean) |
| Lungs | V20 <10% | 4.125 |
| Target | Prescribed dose (Gy) | Mean dose (DVH) (Gy) |
| PTV1 | 24 | 24.51 |
| PTV2 | 27 | 27.19 |
| PTV3 | 30 | 30.74 |
| Homogeneity index (HI) | 1.13 |
For quality assurance, a tighter gamma pass rate was set due to the proximity of the spinal cord and the multiple prescription levels. PSQA volumetric dosimetry was undertaken with an Octavius 4D system (PTW). Evaluated voxel passing rates were 87.8% for 1 mm/3%; 95.8% for 2 mm/2%; and 97.6% for 2 mm/3%. Multi-channel absolute dosimetry measurements showed <3% difference from calculated values (for five ionization chambers), with a maximum dose gradient of 2.8%10.
Treatment delivery
This SIB-SABR plan was delivered in December 2016, with an interval of 40 hours between treatment fractions. A total of 5143.2 MU were delivered for each fraction.
Precise patient positioning with six degrees of freedom was achieved using the HexaPOD evo RT couch. XVI CBCT images were acquired prior to and following each treatment, using in-house presets, and matched to the reference CT simulation images6.
The XVI scanning protocol and treatment delivery beams were optimized so that the XVI scan stopped at 180 degrees and the treatment delivery started at 180 degrees, thereby minimizing the total treatment time.
The workflow for imaging and treatment delivery, including the time in minutes for each step, is shown in Table 2.
Table 2. Treatment delivery workflow
| Step | Procedure | Time (minutes) |
| Pretreatment XVI scan | From kV beam on to reconstruction completed | 1 |
| XVI match | From reconstruction completed to MV beam on for the first arc | 2 |
| Treatment delivery of 4 VMAT arcs (5143.2 MU) | From MV beam on to MV beam off | 10 |
| Post-treatment XVI scan | From kV beam on to reconstruction completed | 1 |
| XVI match | From reconstruction completed to the final assessment | 2 |
Outcome and follow-up
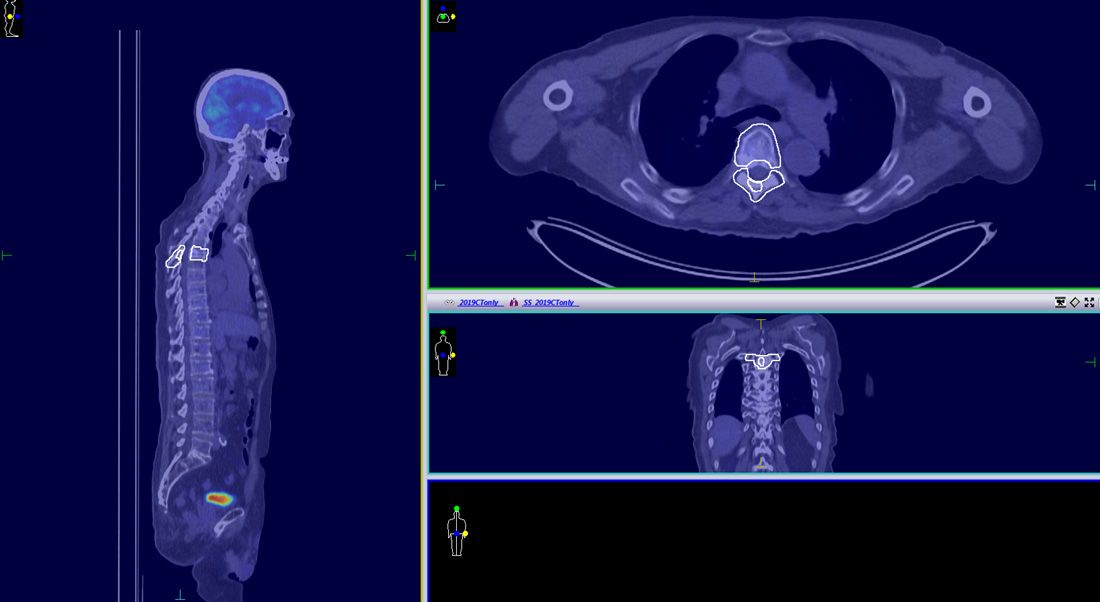
The patient completed vertebral body SABR successfully in 2016 with no side effects observed up to two and half years following treatment. PET–CT examination, performed in October 2019, showed no sign of relapse (Figure 4).
Discussion and conclusions
Elekta Versa HD and Monaco provide a suitable modality for doughnut shaped SABR dose delivery with SIB to a spinal vertebrae, with acceptable spinal cord sparing. PSQA results confirmed that the treatment plan can be prepared and delivered with both very high geometrical precision and resolution of dose distribution.
The planning process (marking-up, constraints, prescriptions, evaluation metrics) were based on SBARCG. The tolerances proposed in this document for PDS and MGI metrics could not be used here for the following reasons: 1. PTV30Gy is placed in PTV27Gy and it is impossible to make sharp gradients to reduce the V100% volume around of PTV30Gy for the overlapping regions with two prescribed dose levels (Figure 3); 2. The PDS (PTV24Gy) is caused by 24 Gy isodose surrounding PTV24Gy and the high dose region volume, which is adjacent to PTV27Gy and PTV30Gy (Figure 3); and 3. PTV30Gy is placed in PTV27Gy and V50% can be bulky in order to provide the higher dose to the internally boosted region (Figure 3). Despite this, V50% still met ICRU recommendations of not expanding to more than 2 cm from PTVs. Acceptability of the plan was assessed on DVH statistics and slice-by-slice reviewing.
References
- Stereotactic Ablative Body Radiotherapy (SABR): A Resource. SABR UK Consortium 2019.
- Planning of SABR spinal vertebrae “doughnut” radiotherapy treatment incorporating an SIB for an Elekta VERSAHD Linear Accelerator. SABR 2019 UK Consortium. Jaganathan A, Khoo V, Fazlic S, Oborska-Kumaszynska D, Weatherburn H.
- The role of radiation therapy in bone metastases management. Oncotarget, 2017, Vol. 8, (No. 15), pp: 25691-25699. De Felice F, et al.
- Can all centers plan intensity modulated radiotherapy (IMRT) effectively? An external audit of dosimetric comparisons between three-dimensional conformal radiotherapy and IMRT for adjuvant chemoradiation for gastric cancer. Int. J. Radiat. Oncol., Biol., Phys. 71(4), 1167–1174 (2008). Chung H, Lee B, Park E, Lu J, and Xia J.
- IMRT treatment planning—a comparative inter-system and inter-centre planning exercise of the ESTRO QUASIMODO group. Radiother. Oncol. 76(3), 354–361 (2005). Bohsung J, Gillis S, Arrans R, Bakai A, de Wagter C, Knöös T, Mijnheer B, Paiusco M, Perrin B, Welleweerd H, and Williams P.
- On target: ensuring geometric accuracy in radiotherapy. The Royal College of Radiologists, Institute of physics and Engineering in Medicine, Society and College of Radiographers. London, UK: The Royal College of Radiologists; 2008.
- A decision making adaptive sandwiching approximation method based MCO approach for SABR spinal vertebrae “doughnut” cases - Monaco Pathway. IPEM's Radiotherapy Special Interest Group- Stereotactic Ablative Radiotherapy 2020. Jaganathan A, Khoo V, Fazlic S, Oborska-Kumaszynska D, Weatherburn H.
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6645671/
- https://www.hindawi.com/journals/bmri/2016/6805979/
- ICRU Report 91 Journal of the International Commission on Radiation Units and Measurements, Volume 14, Issue 2, 2014, Pages 1–160.